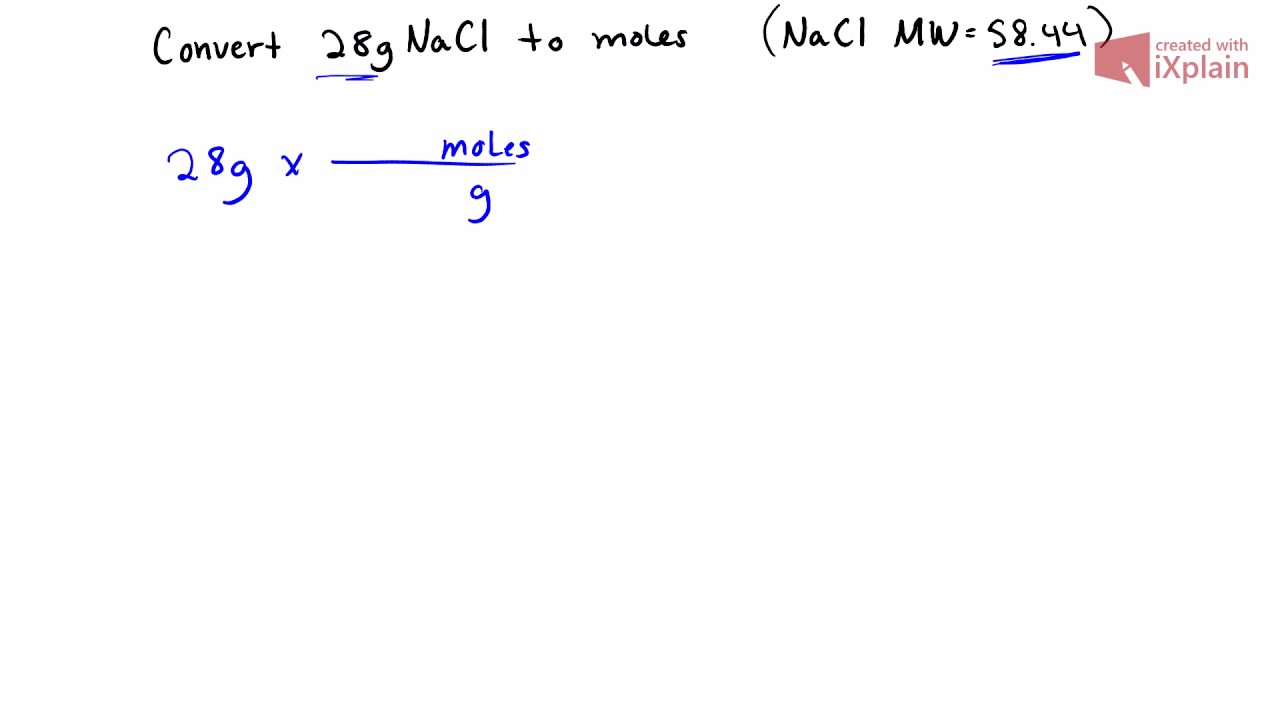Molarity Problems – IF worksheet. 1)MW NaCl = = 58g NaCl = 1 mole 2)M = moles M = x moles Liters 1.0 L 3)58 g NaCl 1 mole NaCl = 1 mole NaCl ppt download

The free energy change when 1 mole of NaCl is dissolved in water at 298 K. is `-x` KJ find out value - YouTube

Explain in why on addition of 1 moe of NaCl to 1L of water, the boiling point of water increases, - YouTube

a) Digital image showing water, NaCl (20 wt%), NaOH (1 mol·L −1 ) and... | Download Scientific Diagram

Calculate the Gibb's energy change when i mole of NaCl is dissolved in water at 25^∘ C. Lattice energy of NaCl = 777.9 KJ mol^- 1 , Δ S for dissolution =
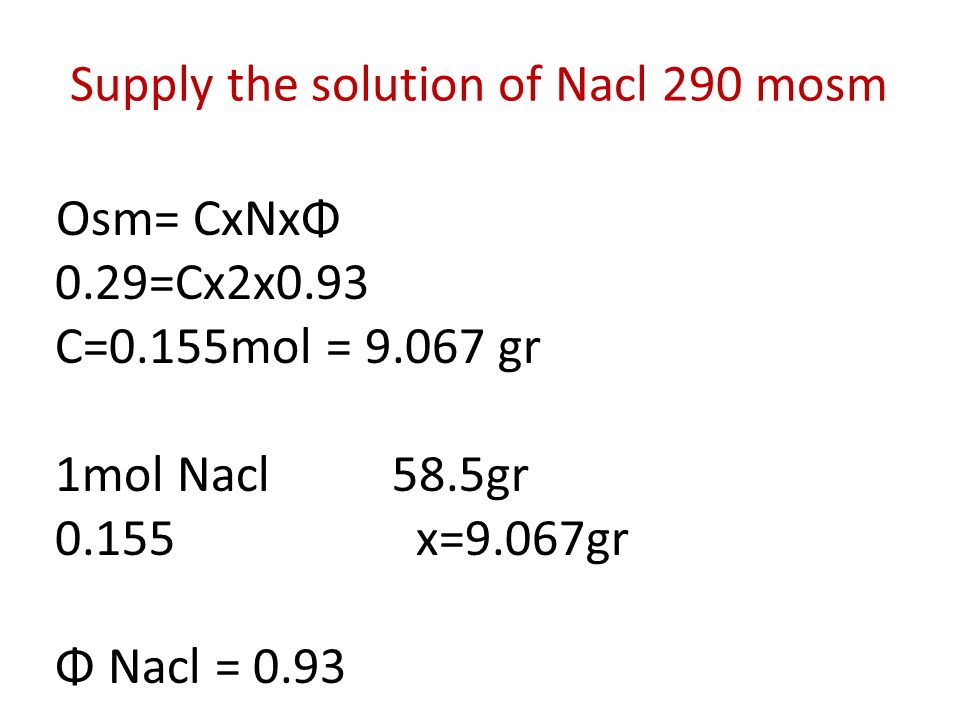
Osmosis 1mol=1moleculgram disolved particle Osmol : 1Osmol =1mol disolved particles 1mol=1moleculgram disolved particle. - ppt download

Effects of 1 mol/L NaCl and the 1 mol/L NaNO3 electrolytes on surface... | Download Scientific Diagram

Concentration Calculations Molarity. Objectives To calculate the molecular weight and moles of a substance To calculate the Molarity of a substance using. - ppt download