Overall reaction of Cathode and Anode: Cathode: 2 H2O(l) + 2e− → H2(g)... | Download Scientific Diagram

SOLVED: When the equation Ca(OH)2 + H3PO4 –> Ca3(PO4)2 + H2O is balanced, the coefficient for H2O is

Complete the following equation: Na2O2 + 2H2O → (W) + H2O2 2KO2 + 2H2O→ (X) + (Y) + O2 Na2O + CO2→ (Z)
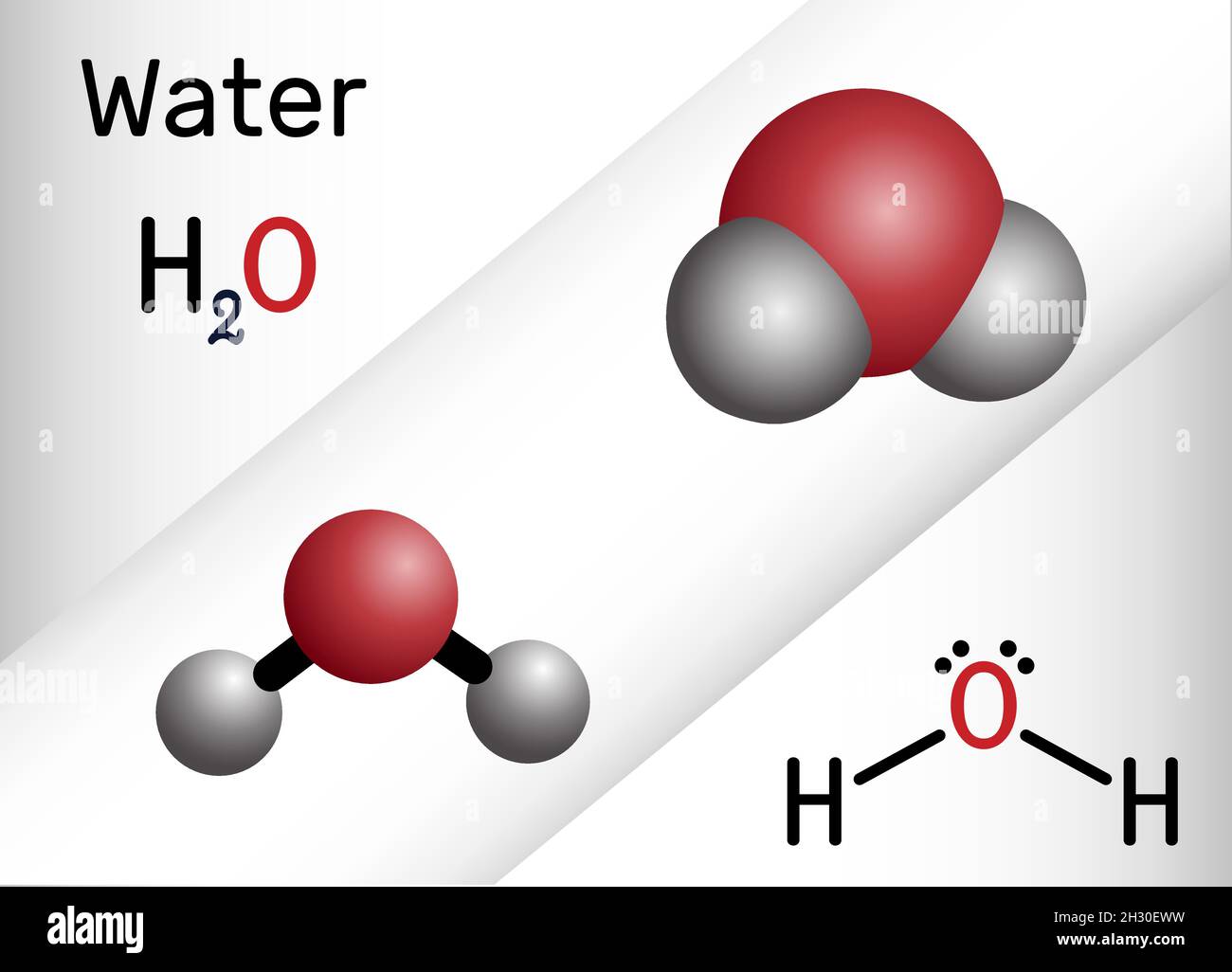
Water , H2O, HOH molecule. It is inorganic hydroxy compound, oxygen hydride consisting of an oxygen atom and two hydrogen atoms. Structural chemical f Stock Vector Image & Art - Alamy
![Milde Hydrothermalsynthese von Na-Cancrinit Na8[AlSiO4]6(NO3)2(H2O)4 bei 60 GradC und Untersuchungen zur thermischen Stabilität (kartoniertes Buch) | Bessunger Buchladen Milde Hydrothermalsynthese von Na-Cancrinit Na8[AlSiO4]6(NO3)2(H2O)4 bei 60 GradC und Untersuchungen zur thermischen Stabilität (kartoniertes Buch) | Bessunger Buchladen](https://medien.umbreitkatalog.de/bildzentrale_original/978/374/480/0952.jpg)
Milde Hydrothermalsynthese von Na-Cancrinit Na8[AlSiO4]6(NO3)2(H2O)4 bei 60 GradC und Untersuchungen zur thermischen Stabilität (kartoniertes Buch) | Bessunger Buchladen

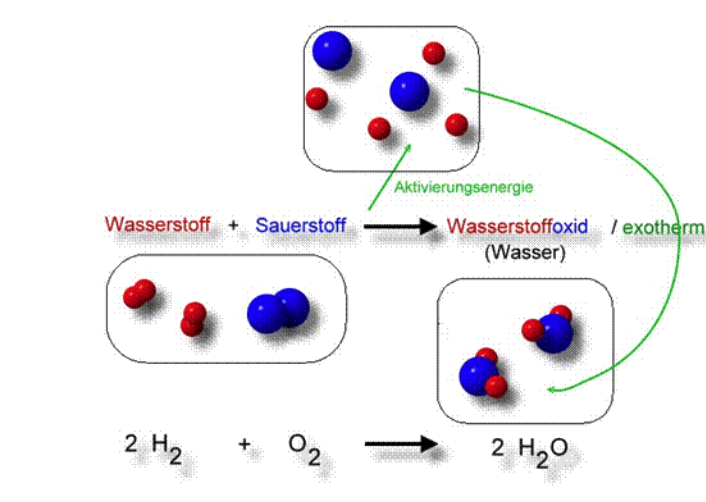
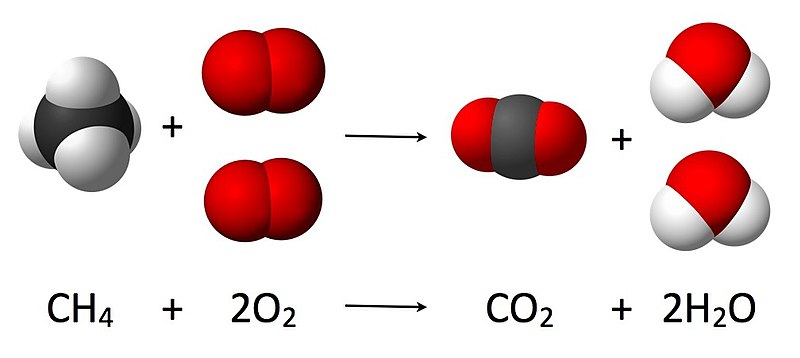
SOLVED: 2. A. Consider the equation, 2H2 + O2 → 2H2O. Here, H2 and O2 are called . Group of answer choices reactants products catalysts modules alkalis B. In chemical equations, the

![Solved [Cl-] low: Oxidation: 2 H2O 02 + 4H+ 4e Reduction: 2 | Chegg.com Solved [Cl-] low: Oxidation: 2 H2O 02 + 4H+ 4e Reduction: 2 | Chegg.com](https://media.cheggcdn.com/media/ac0/ac051806-14e4-4144-a9a5-c6edb1f2a071/php07e7Pp)