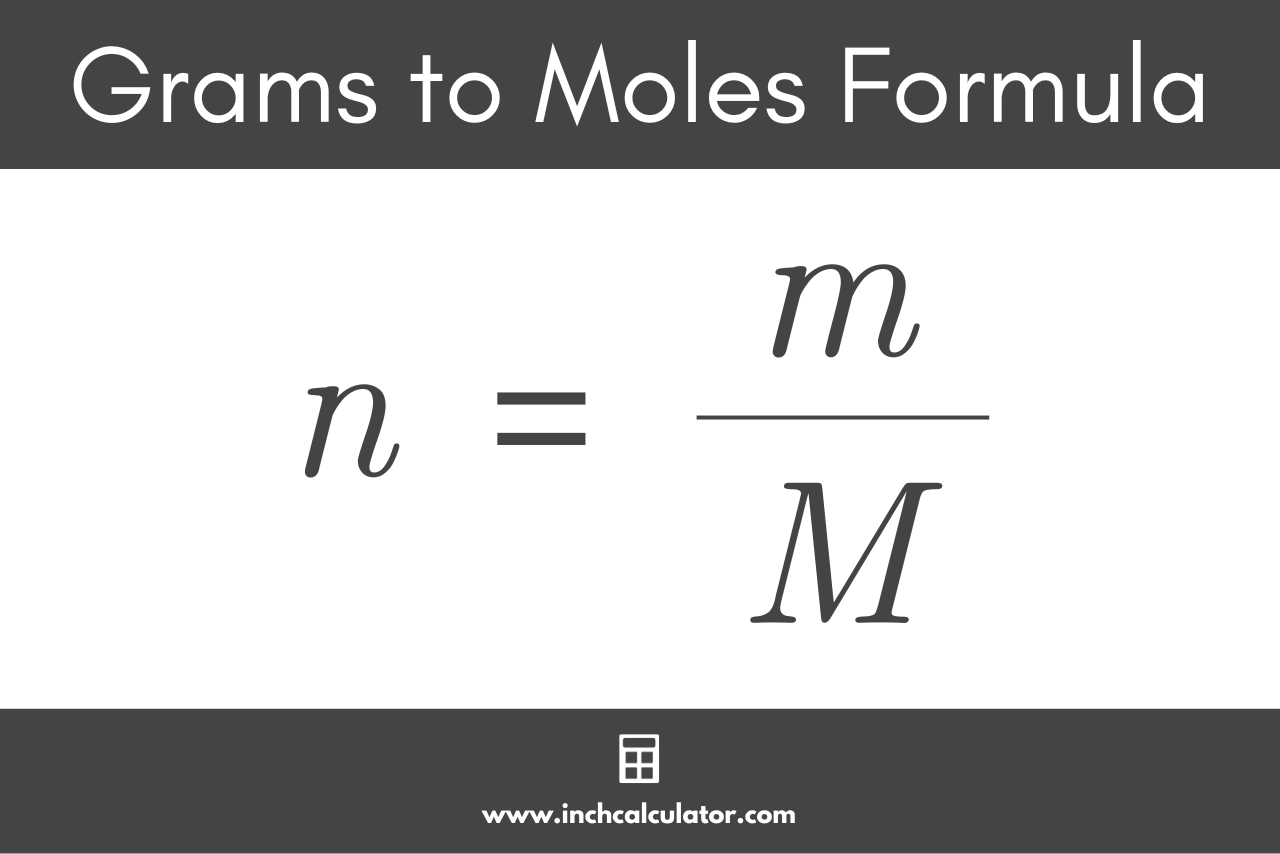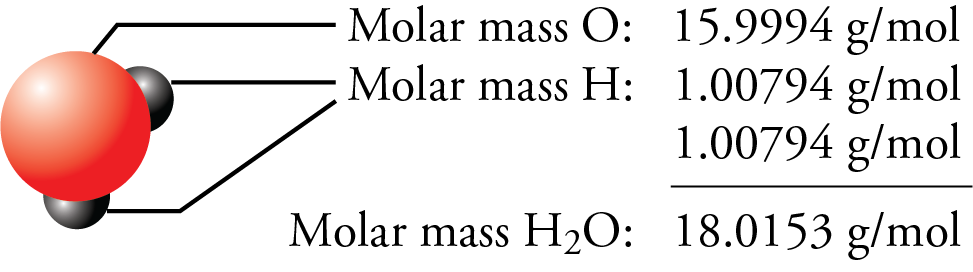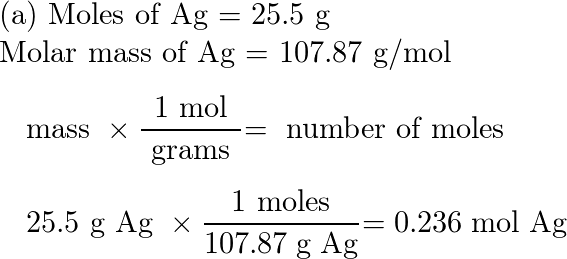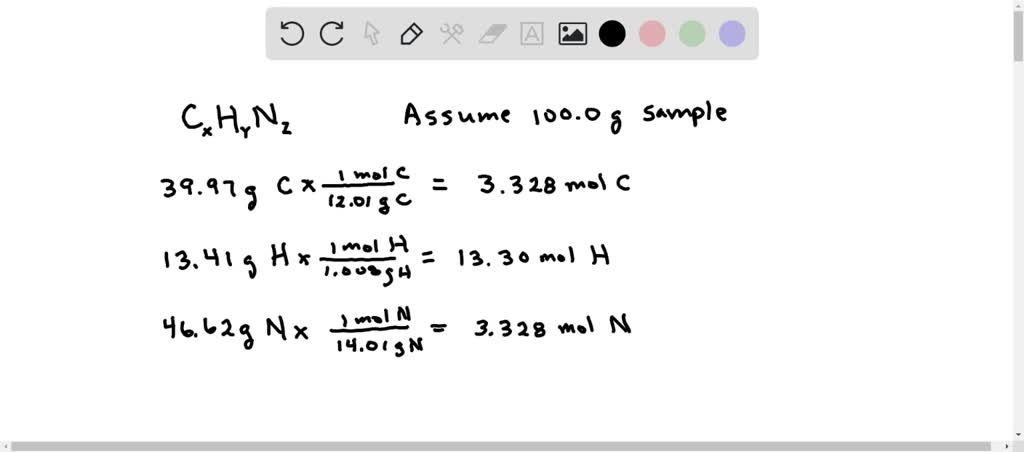
SOLVED: Determine the molecular formula for the compound with a molar mass of 60.10 g/mol and the following percent composition: C, 39.97% H, 13.41% N, 46.62%

Question Video: Determining the Mass of the Potassium Chloride Analyte When 2g of Silver Chloride Precipitates | Nagwa

A Mole is a Unit or have you heard, containing six times ten to the twenty-third That's a six with twenty three zeros at the end, Much to big a number. -
![No. Compound name Probability [%] CAS Number Molecular weight [g mol −1 ] | Download Scientific Diagram No. Compound name Probability [%] CAS Number Molecular weight [g mol −1 ] | Download Scientific Diagram](https://www.researchgate.net/publication/338422297/figure/tbl3/AS:867320046047235@1583796621394/No-Compound-name-Probability-CAS-Number-Molecular-weight-g-mol-1.png)