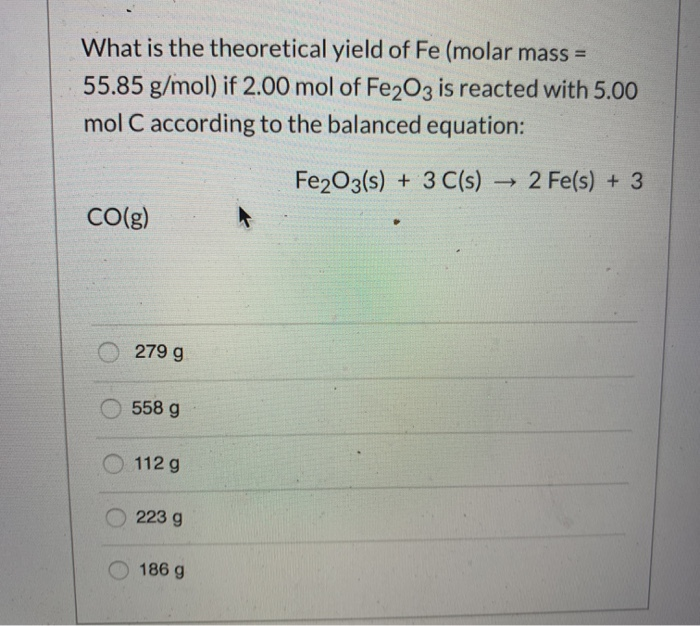
![Ground and Excited States of the [Fe(H2O)6]2+ and [Fe(H2O)6]3+ Clusters: Insight into the Electronic Structure of the [Fe(H2O)6]2+–[Fe(H2O)6]3+ Complex | Journal of Chemical Theory and Computation Ground and Excited States of the [Fe(H2O)6]2+ and [Fe(H2O)6]3+ Clusters: Insight into the Electronic Structure of the [Fe(H2O)6]2+–[Fe(H2O)6]3+ Complex | Journal of Chemical Theory and Computation](https://pubs.acs.org/cms/10.1021/ct501143c/asset/images/medium/ct-2014-01143c_0017.gif)
Ground and Excited States of the [Fe(H2O)6]2+ and [Fe(H2O)6]3+ Clusters: Insight into the Electronic Structure of the [Fe(H2O)6]2+–[Fe(H2O)6]3+ Complex | Journal of Chemical Theory and Computation
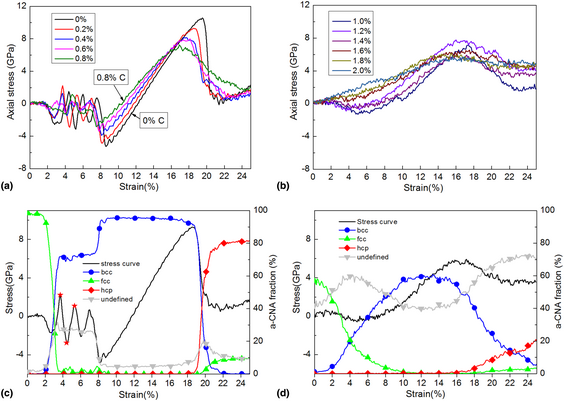
The strain-induced martensitic phase transformation of Fe–C alloys considering C addition: A molecular dynamics study | Journal of Materials Research | Cambridge Core

Fe(deferasirox)2: An Iron(III)-Based Magnetic Resonance Imaging T1 Contrast Agent Endowed with Remarkable Molecular and Functional Characteristics | Journal of the American Chemical Society

Schematic molecular orbital (MO) diagrams of 1 Fe(CO) 4 (left) and 3... | Download Scientific Diagram

Show the full molecular orbital diagram for \begin{bmatrix} Fe(OH_2)_6\end{bmatrix}^{2+}. Indicate all metal atomic orbitals, ligand LGOs, bonding MO's, nonbonding MO's, antibonding MO's, symmetry labels and \Delta_0. | Homework.Study.com

Ferrocene. Molecular model of the organometallic compound ferrocene (C10.H10.Fe). This is a metallocene consisting of two cyclopentadienyl rings bound... - SuperStock
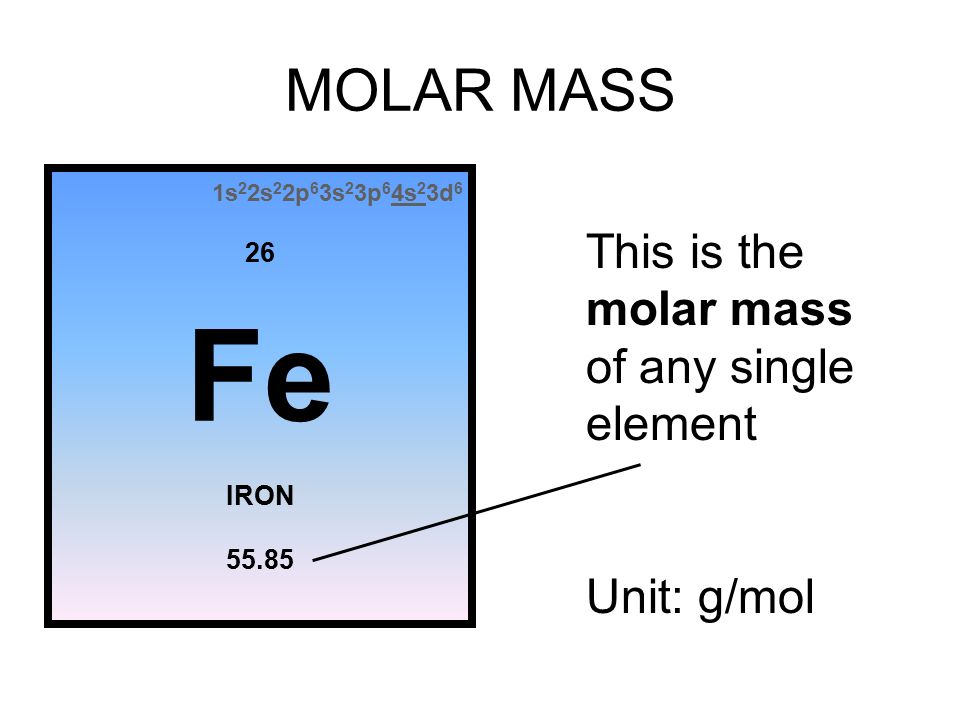
Bling Bling: if I were to give each of you one atom of gold for every second that has elapsed since the Dinosaur's went extinct 65 million years ago, how. - ppt download

If 3.50 mol of Fe reacts with 3.00 mol of oxygen in the following reaction: 4 Fe(s) + 3 O2(g) → 2 - Brainly.com

![ANSWERED] Calculate the molar mass of Fe 3(PO 4) 2... - Inorganic Chemistry ANSWERED] Calculate the molar mass of Fe 3(PO 4) 2... - Inorganic Chemistry](https://media.kunduz.com/media/sug-question/raw/63334580-1657459641.4436913.jpeg)