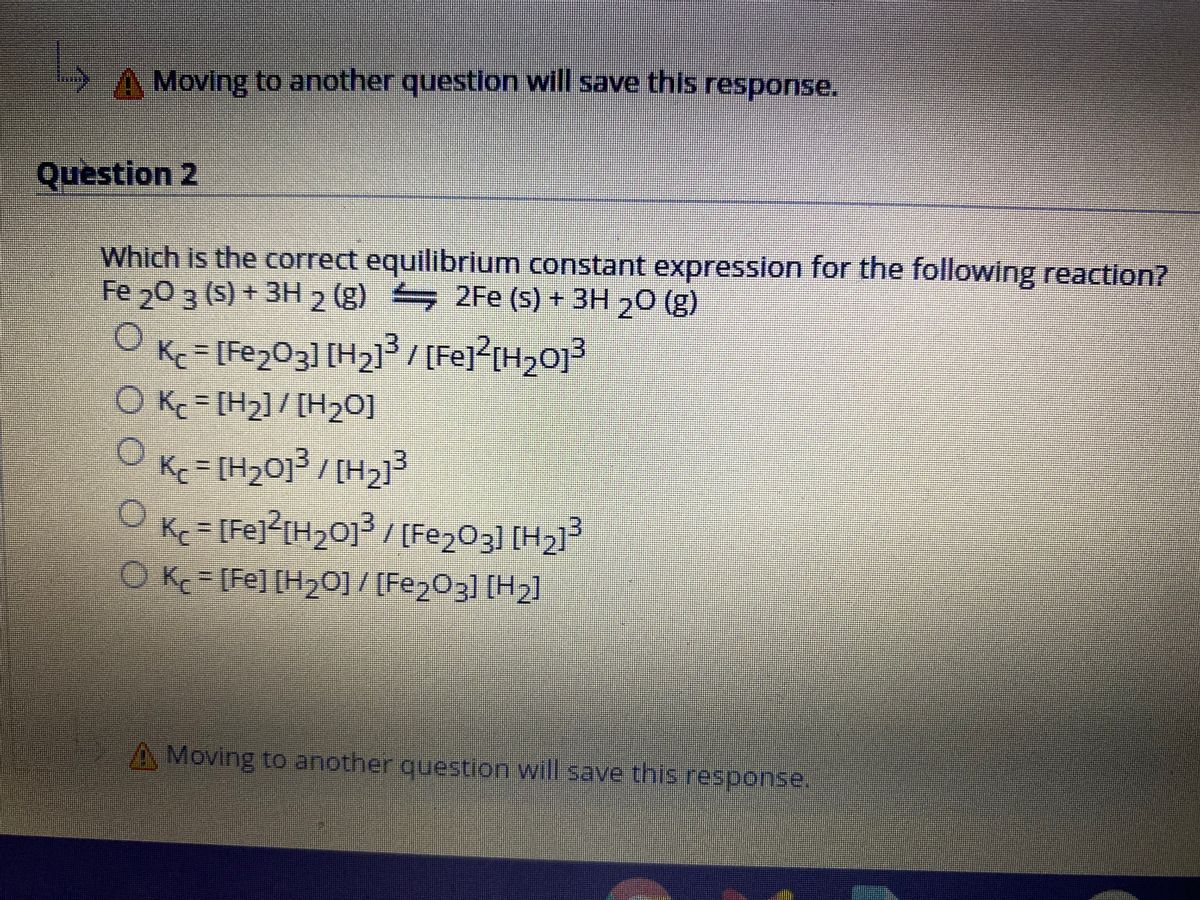
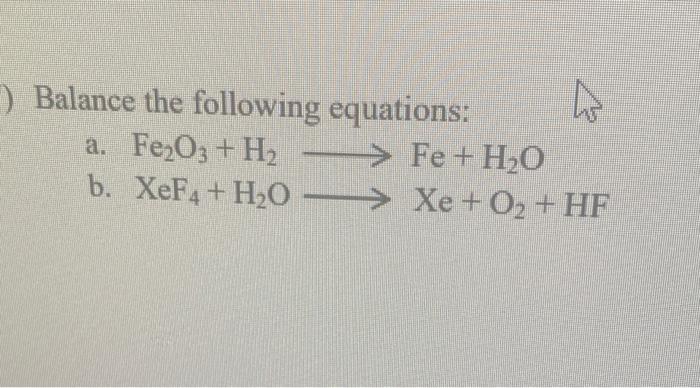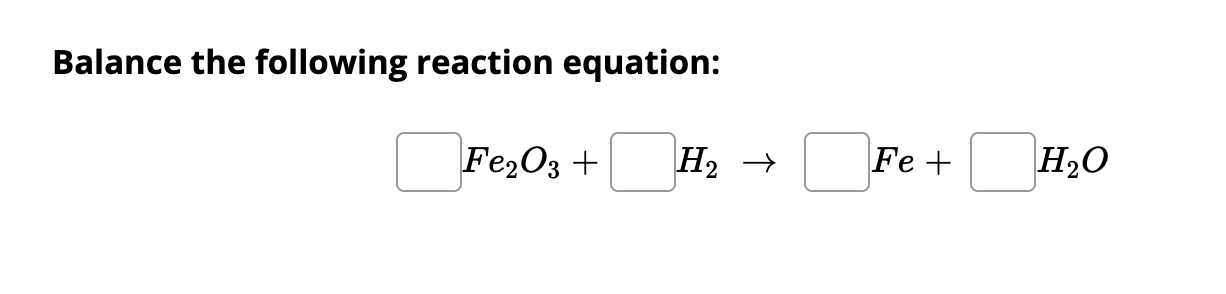PPT - Friday, Aug. 30 th : “A” Day Tuesday, Sept. 3 rd : “B” Day Agenda PowerPoint Presentation - ID:3916169

Balance the equation- rn Fe + O2 + H2O ---- Fe2O3 - Science - Physical and Chemical Changes - 11497 | Meritnation.com

SOLVED: Using the following equation: Fe2O3 + H2 –> Fe + H2O Calculate how many grams of iron (Fe) can be made from 16.5 grams of Fe2O3 from the chemical equation. PLEASE SHOW WORK!!!