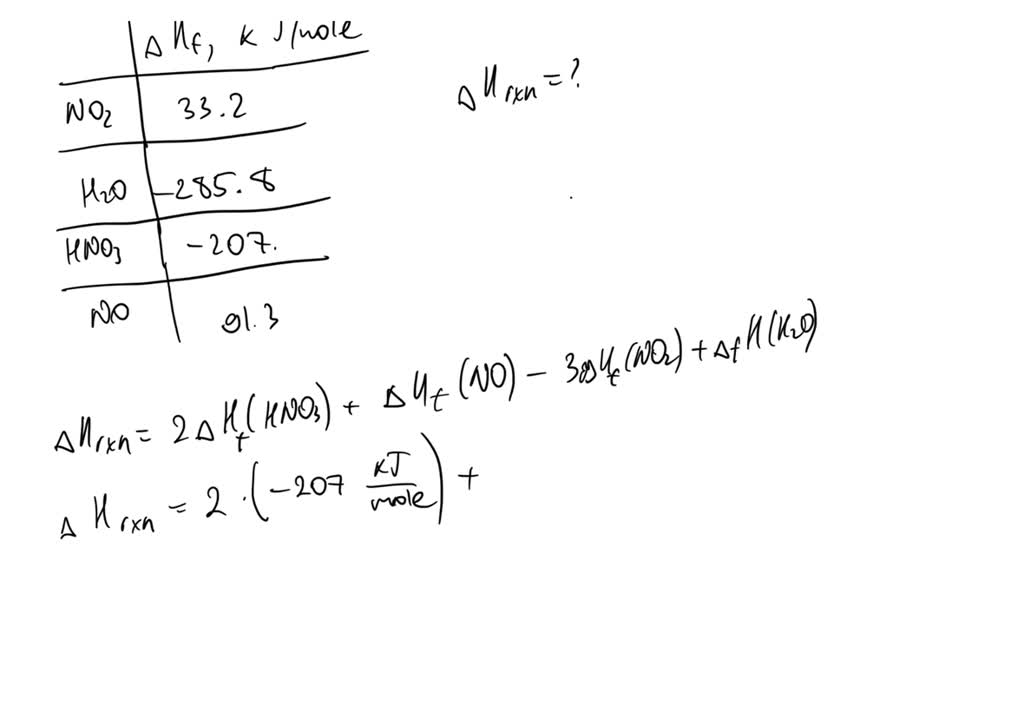The reaction, CO (g) + 3H2 (g) ⇌ CH4 (g) + H2O (g)is at equilibrium at 1300 K in a 1L flask..... - YouTube

Water decomposition yield, G(-H2O), has a function of the flux ratio... | Download Scientific Diagram

The Δ Hf^0 for CO2 (g), CO(g) and H2O(g) are - 393.5, - 110.5 and - 241.8 kJ mol^-1 respectively. The standard enthalpy change (in kJ) for the reaction CO2 (g) +

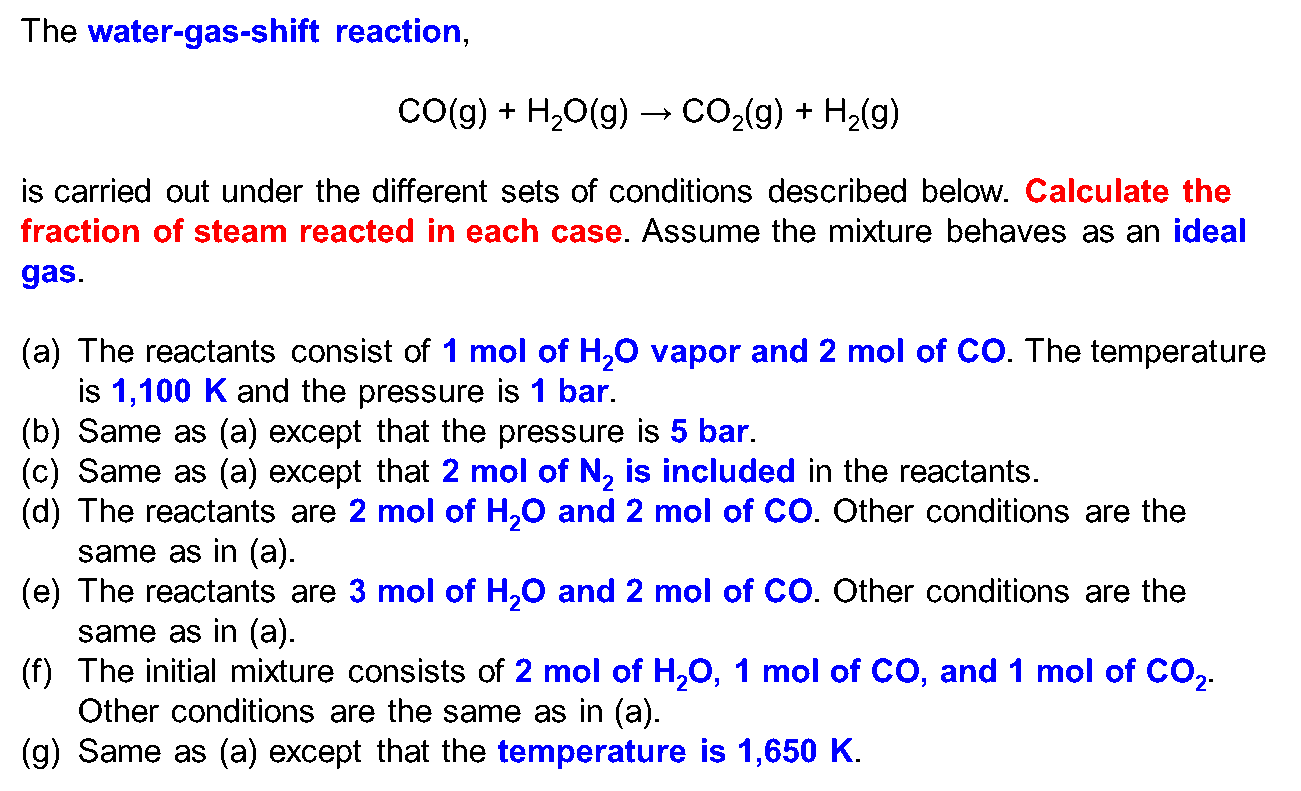
OneClass: 4) For the reaction H2(g) + CO2(g) H2O(g) + CO(g) at 700°C, Kp = 0.534. Calculate the numb...

See: Calculate the amount of heat released when 27.0 g H2O is cooled from a liquid at 314 K to a solid at - Brainly.com