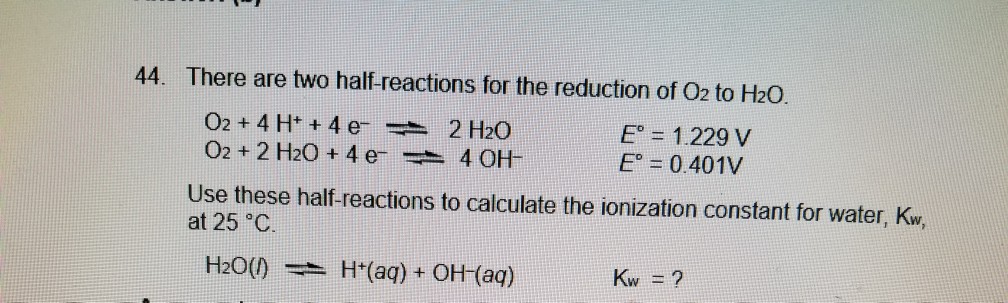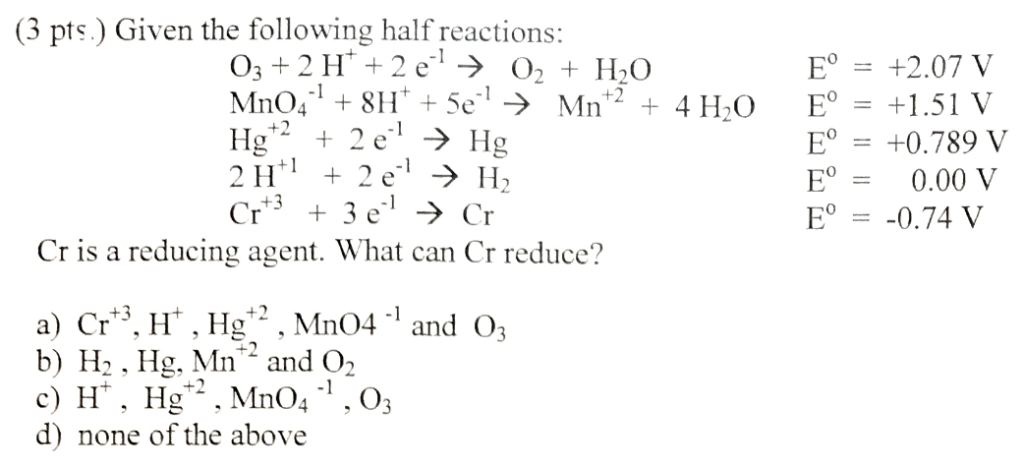The standard reduction potential of the reaction at 25^° C ,Н2О + e^ gives half H2(g) +OH^ is 0.8277 V. The equilibrium constant for the reaction H2O reversible H3O^++OH^ at 25^° C is

SOLVED: the transfer of electrons in the respiratory chain in the mitochondria can be lrepresented by the reaction: NADH +H(+) 02 -> H2o NAD (+) A) Calculate delta E for the reaction

Reduction of O2 to H2O and its free radical intermediates (A) Lewis... | Download Scientific Diagram



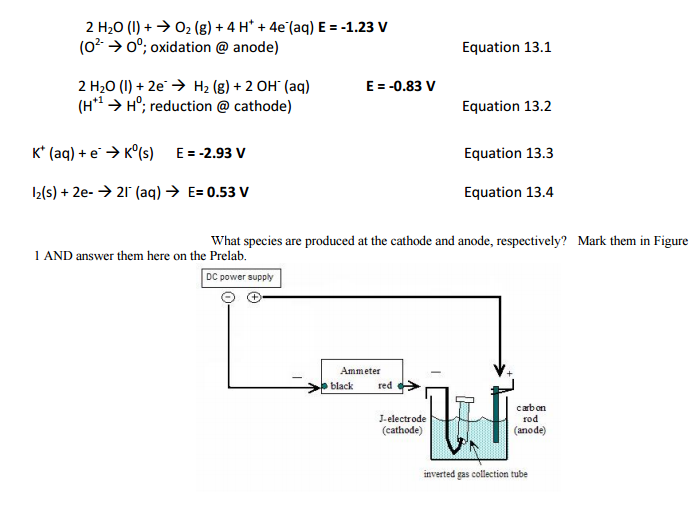







![Solved [Cl-] low: Oxidation: 2 H2O 02 + 4H+ 4e Reduction: 2 | Chegg.com Solved [Cl-] low: Oxidation: 2 H2O 02 + 4H+ 4e Reduction: 2 | Chegg.com](https://media.cheggcdn.com/media/ac0/ac051806-14e4-4144-a9a5-c6edb1f2a071/php07e7Pp)