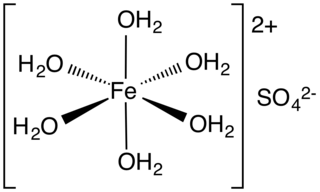
SOLVED: Balance the following redox reaction if it occurs in H2SO4. What are the coefficients in front of H2O and Cr2(SO4)3 in the balanced reaction? C6H14O2(aq) + K2Cr2O7(aq) → C6H10O4(aq) + Cr2(SO4)3(aq)

7788-99-0 | Chromium Potassium Sulfate Dodecahydrate | Chromium Potassium Sulfate (KCr(SO4)2) Dodecahydrate; Monopotassium Monochromium Disulfate Dodecahydrate; Potassium Chromium Disulfate Dodecahydrate; | CrK(SO4)2 • 12 (H2O) | TRC
![Y 3+ is coordinated by nine oxygen atoms in NaY[SO4]2 • H2O with C2... | Download Scientific Diagram Y 3+ is coordinated by nine oxygen atoms in NaY[SO4]2 • H2O with C2... | Download Scientific Diagram](https://www.researchgate.net/publication/351749785/figure/fig2/AS:1027001976635392@1621867760325/Y-3-is-coordinated-by-nine-oxygen-atoms-in-NaYSO42-H2O-with-C2-symmetry-left-and.png)
Y 3+ is coordinated by nine oxygen atoms in NaY[SO4]2 • H2O with C2... | Download Scientific Diagram
How many unpaired electrons are present in the Brown Ring complex [Fe(H2O )5(NO)]SO4 (1) 4 (2) 3 (3) 0 (4) 5

Balance the following chemical reactions (i) Fe + H2SO4 → Fe2(SO4)3 + H2 (ii) C2H6 + O2 → H2O - Science - Chemical Reactions and Equations - 16678311 | Meritnation.com

How to Balance Al2O3 + H2SO4 = Al2(SO4)3 + H2O,How to Balance Aluminum oxide + Sulfuric acid,How to Balance Aluminum oxide + Sulfuric acid + Aluminum sulfate + Water,balancing Al2O3 + H2SO4 =

H2O+H2SO4=H3O+SO4 Balance the equation. h2o+h2so4=h3o+so4 water and Sulfuric acid reacts to form - YouTube
![SOLVED: [Cu(NH3)4]SO4 • H20 (s) was prepared from 15.5 grams CuSO4 • 5 H2O (s) using a total of 10 mL of 15 M NH3. Determine which reactant is limiting. Molar Masses: SOLVED: [Cu(NH3)4]SO4 • H20 (s) was prepared from 15.5 grams CuSO4 • 5 H2O (s) using a total of 10 mL of 15 M NH3. Determine which reactant is limiting. Molar Masses:](https://cdn.numerade.com/ask_previews/3e5daa89-75dc-40aa-92e7-425d1e093c86_large.jpg)




![Answered: a. CГОЗ HO H,SO4, H2O b. [1] O3 [2] H20… | bartleby Answered: a. CГОЗ HO H,SO4, H2O b. [1] O3 [2] H20… | bartleby](https://content.bartleby.com/qna-images/question/d66f1a49-6a74-4120-82f6-39c04434f2ae/0caae1aa-a18d-4475-b945-b3b8c68a132a/nw3bhka.png)