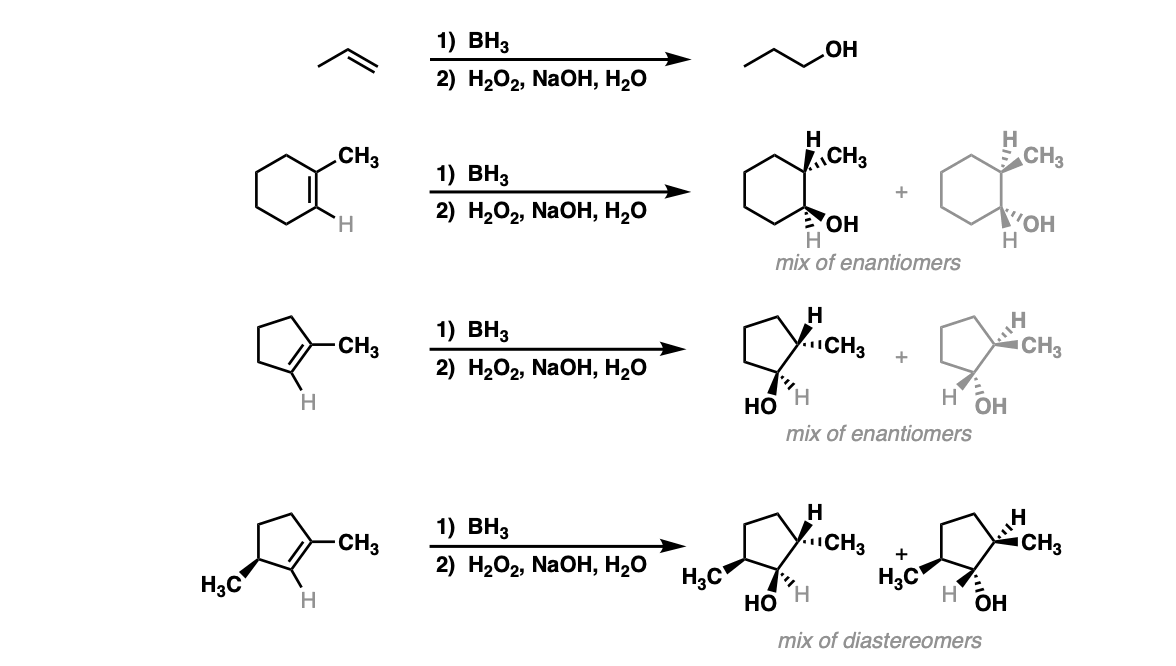
Why is the answer B? Can someone explain this to me and why other options are incorrect. I assumed that H2O2 will decompose rapidly to form H20 and O2 with MnO2 as

Kinetic studies on the reaction of 2 with H2O2 in buffered MeCN/H2O... | Download Scientific Diagram

Direct production of H2O2 from H2 and O2 in a biphasic H2O/scCO2 system over a Pd/C catalyst: Optimization of reaction conditions - ScienceDirect

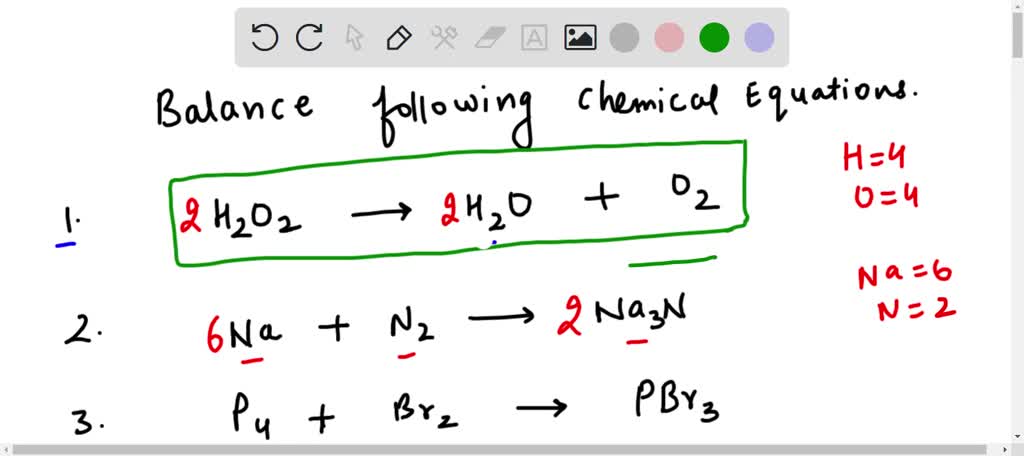
SOLVED: Balance the following equation: H2O2 HzO + 02 H202 HzO + 02 2 H2O2 2 HzO + 02 2 HzO2 2 H2O + 302 H2Oz 2 HzO + 02
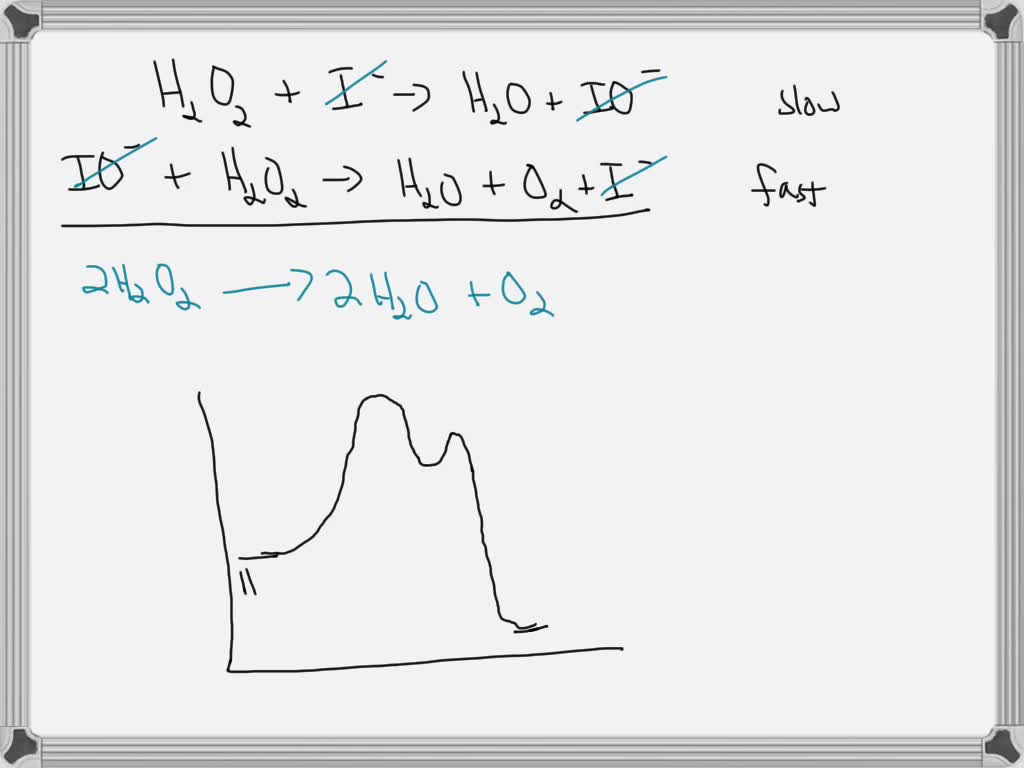
SOLVED: Step 1: H2O2 + I- → H2O + IO- (slow) Step 2: H2O2 + IO- → H2O + O2 + I- (fast) a.) Identify the rate determining step. b.) Draw a

Reduction of O2 to H2O and its free radical intermediates (A) Lewis... | Download Scientific Diagram
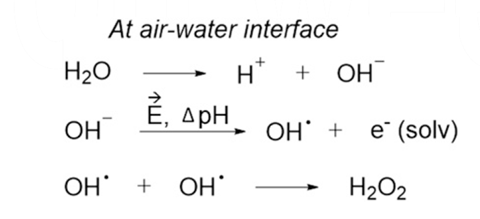


![For the reaction ; 2H2O2(aq)→ 2H2O(l) + O2(g) , rate of decomposition for H2O2 = k[H2O2]^2 For the reaction ; 2H2O2(aq)→ 2H2O(l) + O2(g) , rate of decomposition for H2O2 = k[H2O2]^2](https://dwes9vv9u0550.cloudfront.net/images/2785739/3a8762db-6023-4a5c-9588-862513a5c19d.jpg)