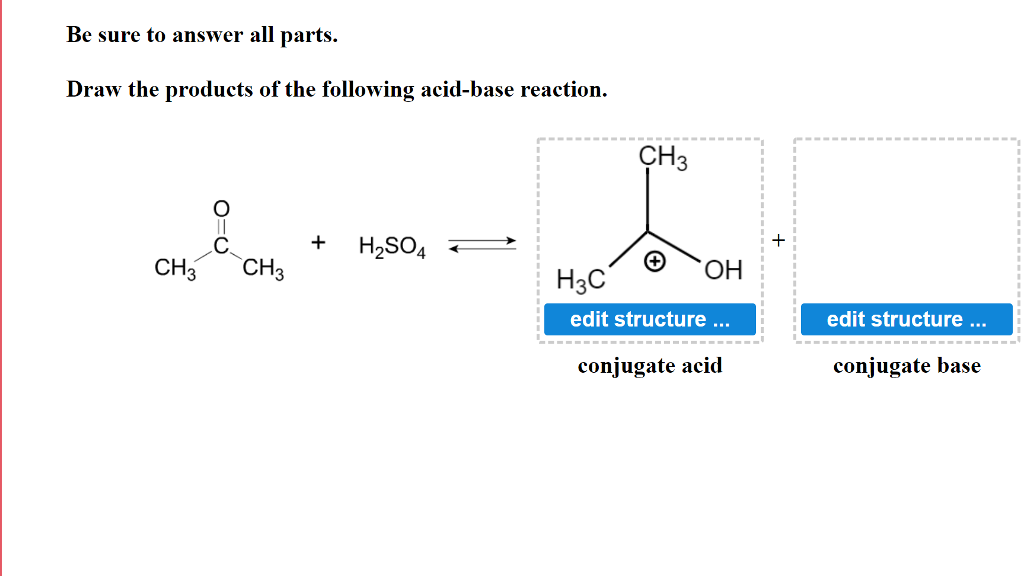
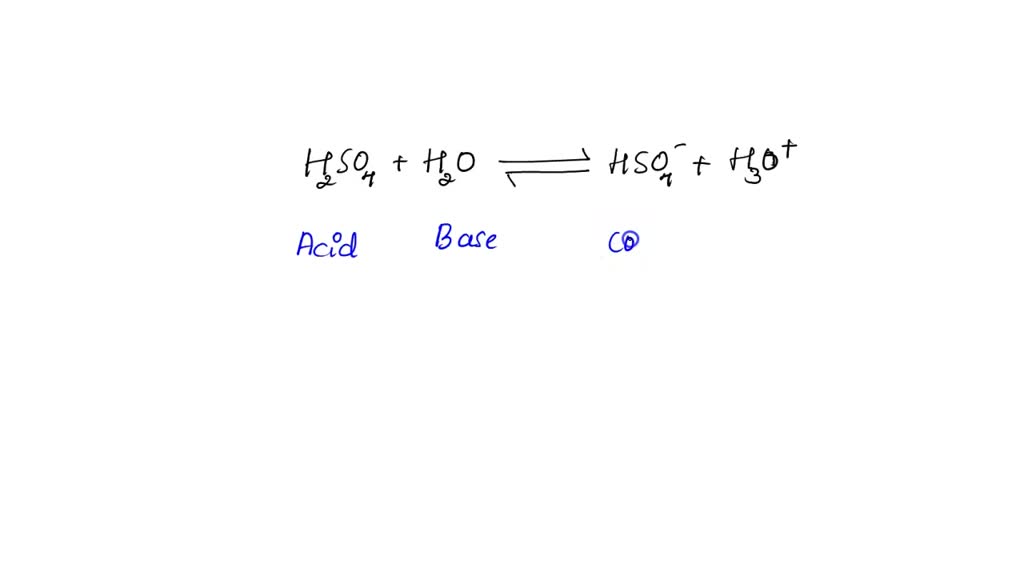Identify the acid-conjugate base pair in this balanced equation: H2SO4 + 2NaOH → 2H2O + - Brainly.com

Illustration of the initial acid‐base clustering in urban Beijing. (a)... | Download Scientific Diagram

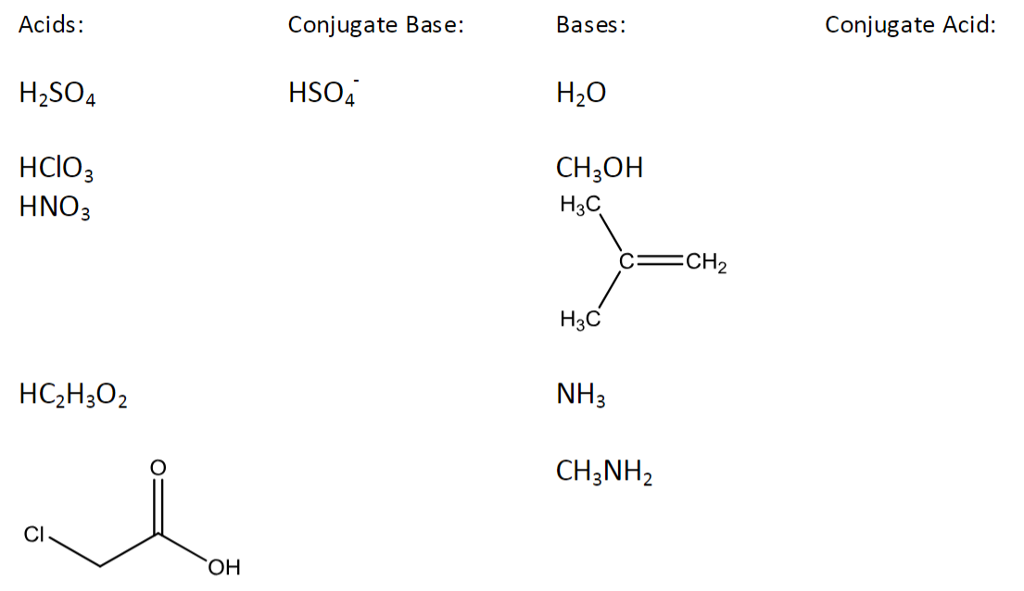
Use your understanding of molecular structure to explain why the conjugate bases of acids like formic acid CHOOH, acetic acid CH3COOH, and phosphoric acid are only stable enough to be weak acids;