
Alumminum hydroxide reacts with sulfuric acid as follows: 2Al(OH)3+H2SO4-->Al2(SO4)+6H2O. Which reagent is the limiting reactant when 0.500 mol Al(OH)3 and 0.500 mol H2SO4 are allowed to react? How ma | Homework.Study.com
Sulfuric acid (sulphuric acid) is a highly corrosive strong mineral acid with the molecular formula H2SO4. It is a pungent-ethereal, colorless to slig Stock Photo - Alamy

The molecular mass of H2SO4 is 98 amu. Calculate the number of moles of each elements in 294 g of H2SO4
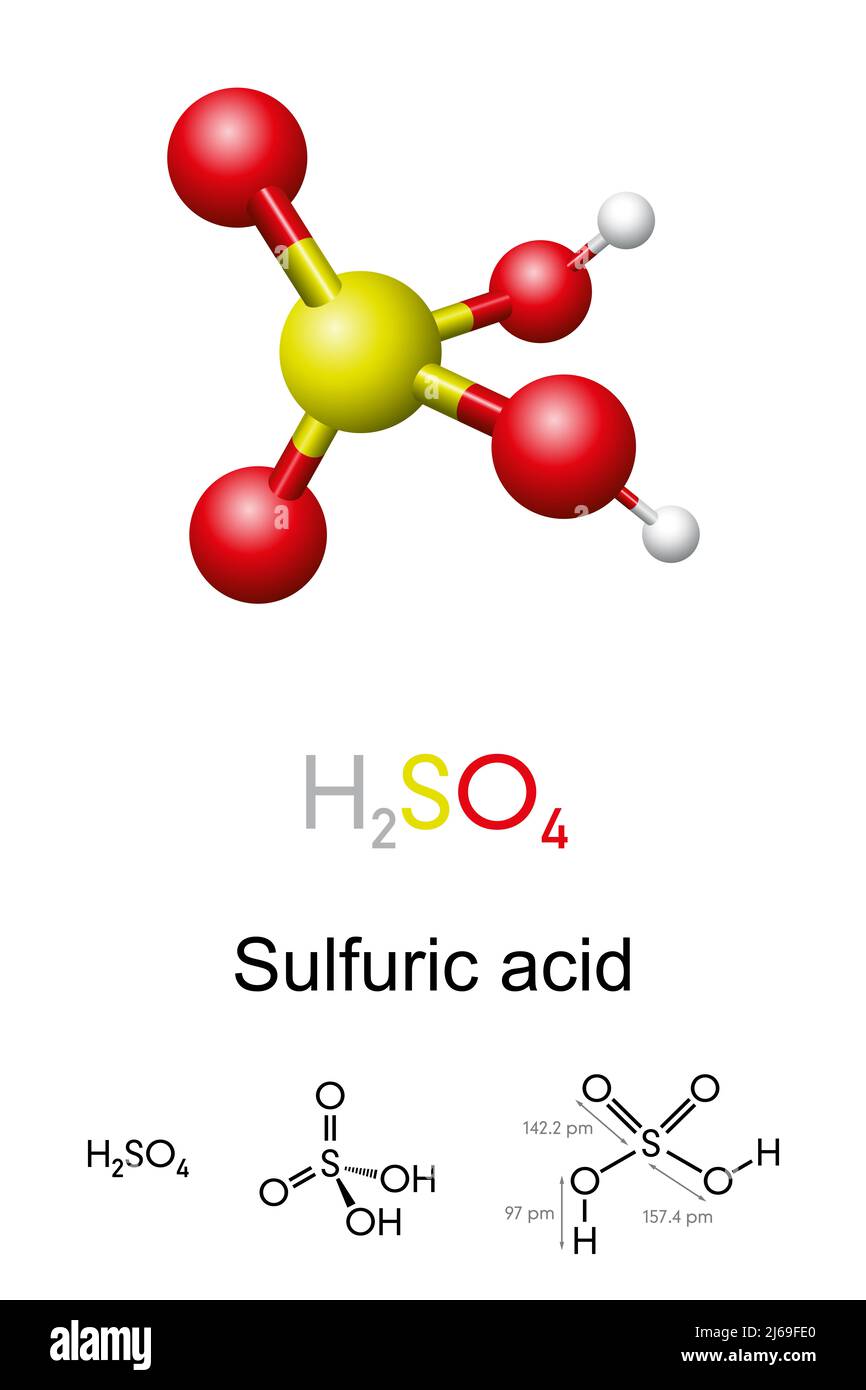
Sulfuric acid, H2SO4, ball-and-stick model, molecular and chemical formula with binding lengths. Known as sulphuric acid, or oil of vitriol Stock Photo - Alamy
100 ml of 3 mol h2so4 reacts with 100 ml of 3 mol naoh. enthalpy of neutralisation of reaction will be 1) 57.1 kJ/ mol 2) 2 × 57.1 kJ 3) 0.3×57.1 kJ 4) 3×57.1 k



![Sulfuric Acid [H2SO4] Molecular Weight Calculation - Laboratory Notes Sulfuric Acid [H2SO4] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2021/11/sulfuric-acid-molecular-weight-calculation.jpg)







