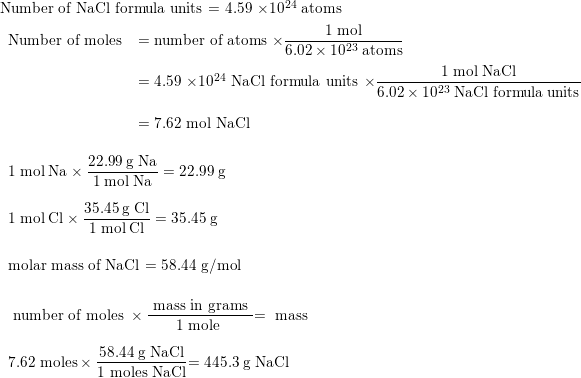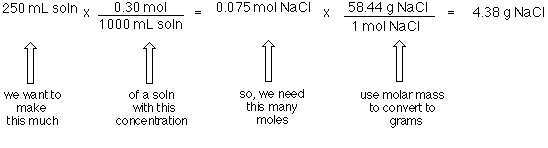3/4/2016 I ObjectiveDo Now Convert grams of a substance to moles of a substance. Calculate the molar mass of: NaCl MgCl ppt download

Effects of 1 mol/L NaCl and the 1 mol/L NaNO3 electrolytes on surface... | Download Scientific Diagram

The Mole & Chemical Quantities. The Mole Mole-the number of particles equal to the number of atoms in exactly 12.0 grams of carbon mol = 6.02 x. - ppt download

The mole fraction of NaCl in an aqueous solution is 0.132. What is the weight/weight percent of NaCl - Brainly.com

Formula mass of NaCl is 58.45 g mol ^-1 and density of its pure form is 2.167 g cm ^-3 . The average distance between adjacent sodium and chloride ions in the

Calculate the mass of 1 mole of each one of the following: (a) `NaCl` , (b) `CaCO_(3)` , (c ) `FeSO - YouTube

Concentration Calculations Molarity. Objectives To calculate the molecular weight and moles of a substance To calculate the Molarity of a substance using. - ppt download

Calculate the Gibb's energy change when i mole of NaCl is dissolved in water at 25^∘ C. Lattice energy of NaCl = 777.9 KJ mol^- 1 , Δ S for dissolution =

Changes of phenol concentration with time for I NaCl = 1 mol kg −1 (a)... | Download Scientific Diagram
Natriumchlorid, 1 kg, Kunst., CAS No. 7647-14-5 | Natriumsalze, Na | Salze zur Analyse (p.a.) | Salze | Anorganische & analytische Reagenzien | Chemikalien | Carl Roth - Deutschland