
Role of Pt and PtO2 in the Oxygen-Evolution Reaction in the Presence of Iron under Alkaline Conditions | Inorganic Chemistry

Adsorption and Dissociation of O2 on Pt−Co and Pt−Fe Alloys | Journal of the American Chemical Society
![PDF] New Pd-Pt and Pd-Au catalysts for an efficient synthesis of H2O2 from H2 and O2 under very mild conditions | Semantic Scholar PDF] New Pd-Pt and Pd-Au catalysts for an efficient synthesis of H2O2 from H2 and O2 under very mild conditions | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b6b16d7b080f74bbb2b2b4709f7613f825e59bab/2-Figure1-1.png)
PDF] New Pd-Pt and Pd-Au catalysts for an efficient synthesis of H2O2 from H2 and O2 under very mild conditions | Semantic Scholar

Dissociative adsorption of O2 on strained Pt(111) - Physical Chemistry Chemical Physics (RSC Publishing)

Catalytic oxidation of CO over Pt/Fe3O4 catalysts: Tuning O2 activation and CO adsorption | SpringerLink

High-Concentration Single Atomic Pt Sites on Hollow CuSx for Selective O2 Reduction to H2O2 in Acid Solution - ScienceDirect

Hydrogen Peroxide Synthesis via Enhanced Two-Electron Oxygen Reduction Pathway on Carbon-Coated Pt Surface | The Journal of Physical Chemistry C

A-C) The schematic atom models of O2 was adsorbed on the surface of... | Download Scientific Diagram
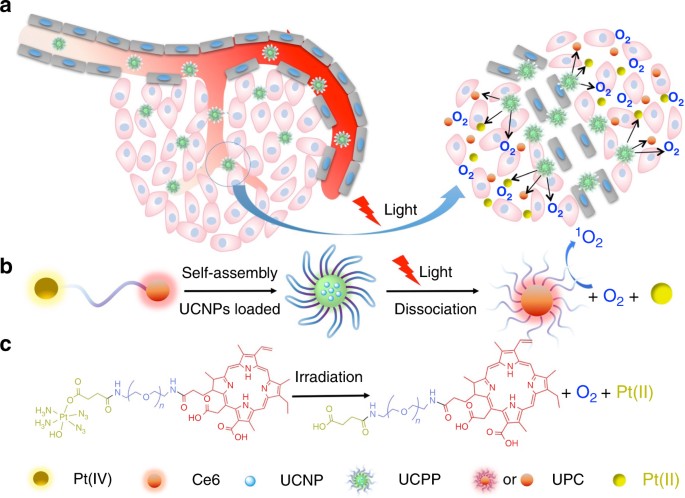
Oxygen and Pt(II) self-generating conjugate for synergistic photo-chemo therapy of hypoxic tumor | Nature Communications
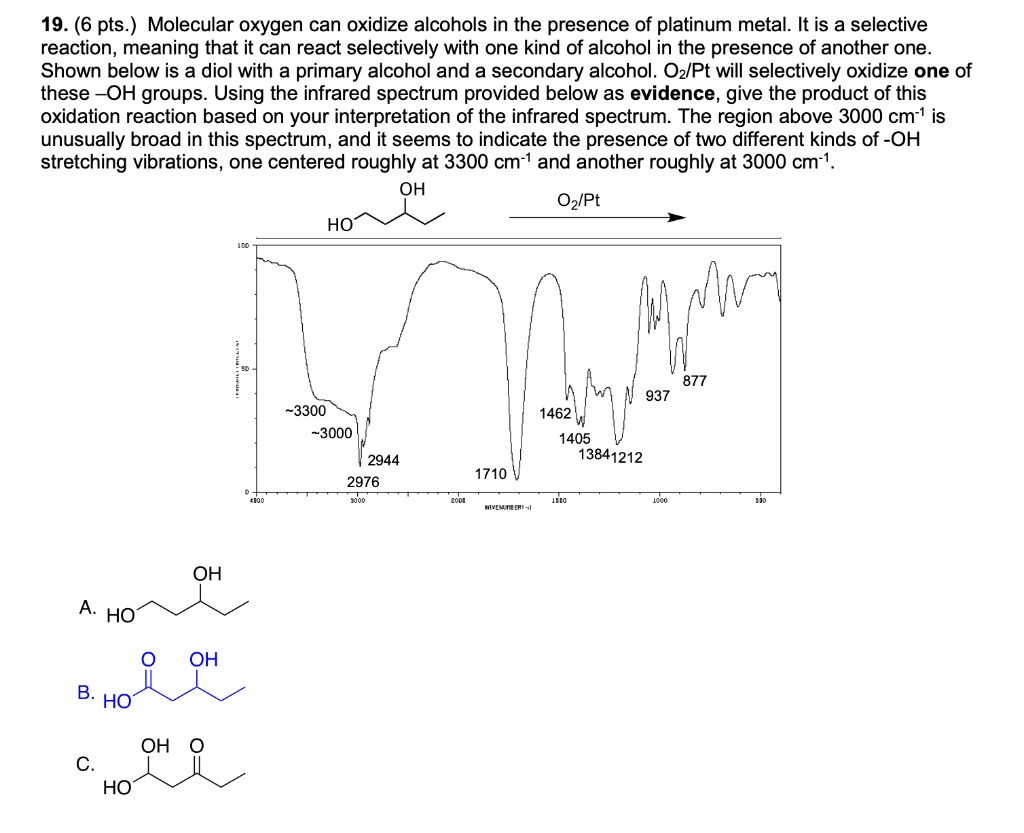
SOLVED: 19. (6 pts Molecular oxygen can oxidize alcohols in the presence of platinum metal. It is a selective reaction, meaning that it can react selectively with one kind of alcohol in
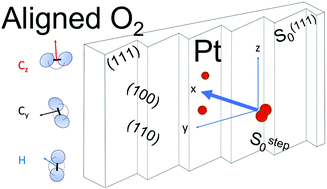
Alignment and impact angular dependence to O2 sticking and dissociation on Pt(111) and close-packed steps - Physical Chemistry Chemical Physics (RSC Publishing)

Theoretical insights into the activation of O2 by Pt single atom and Pt4 nanocluster on functionalized graphene support: Critical role of Pt positive polarized charges - ScienceDirect







