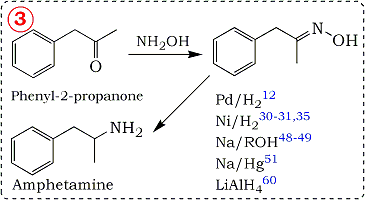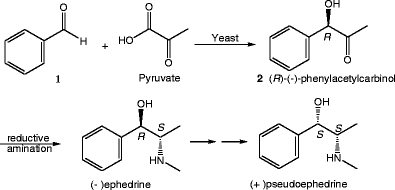
Potential of some yeast strains in the stereoselective synthesis of (R)-(−)- phenylacetylcarbinol and (S)-(+)-phenylacetylcarbinol and their reduced 1,2-dialcohol derivatives | SpringerLink

THE BIOPRODUCTION OF L-PHENYLACETYLCARBINOL USING SOLID-LIQUID TWO PHASE PARTITIONING BIOREACTORS | Semantic Scholar

Improvement of the yeast based (R)-phenylacetylcarbinol production process via reduction of by-product formation - ScienceDirect

Mechanistic study of the biosynthesis of R-phenylacetylcarbinol by acetohydroxyacid synthase enzyme using hybrid quantum mechanics/molecular mechanics simulations - ScienceDirect

Validation of mathematical model with phosphate activation effect by batch (R)-phenylacetylcarbinol biotransformation process utilizing Candida tropicalis pyruvate decarboxylase in phosphate buffer | Scientific Reports
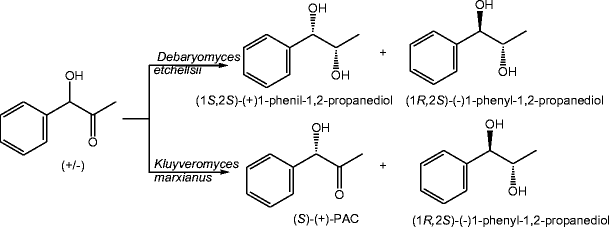
Potential of some yeast strains in the stereoselective synthesis of (R)-(−)- phenylacetylcarbinol and (S)-(+)-phenylacetylcarbinol and their reduced 1,2-dialcohol derivatives | SpringerLink

Asymmetric synthesis of ( S )-phenylacetylcarbinol – closing a gap in C–C bond formation - Green Chemistry (RSC Publishing) DOI:10.1039/C6GC01803C

Investigation of the l-phenylacetylcarbinol process to substituted benzaldehydes of interest - ScienceDirect

Investigation of the l-phenylacetylcarbinol process to substituted benzaldehydes of interest - ScienceDirect